At the end of the 24-week treatment period, 30% of Opzelura patients had at least 75% improvement in the facial Vitiligo Area Scoring Index, compared with 10% of placebo patients. 
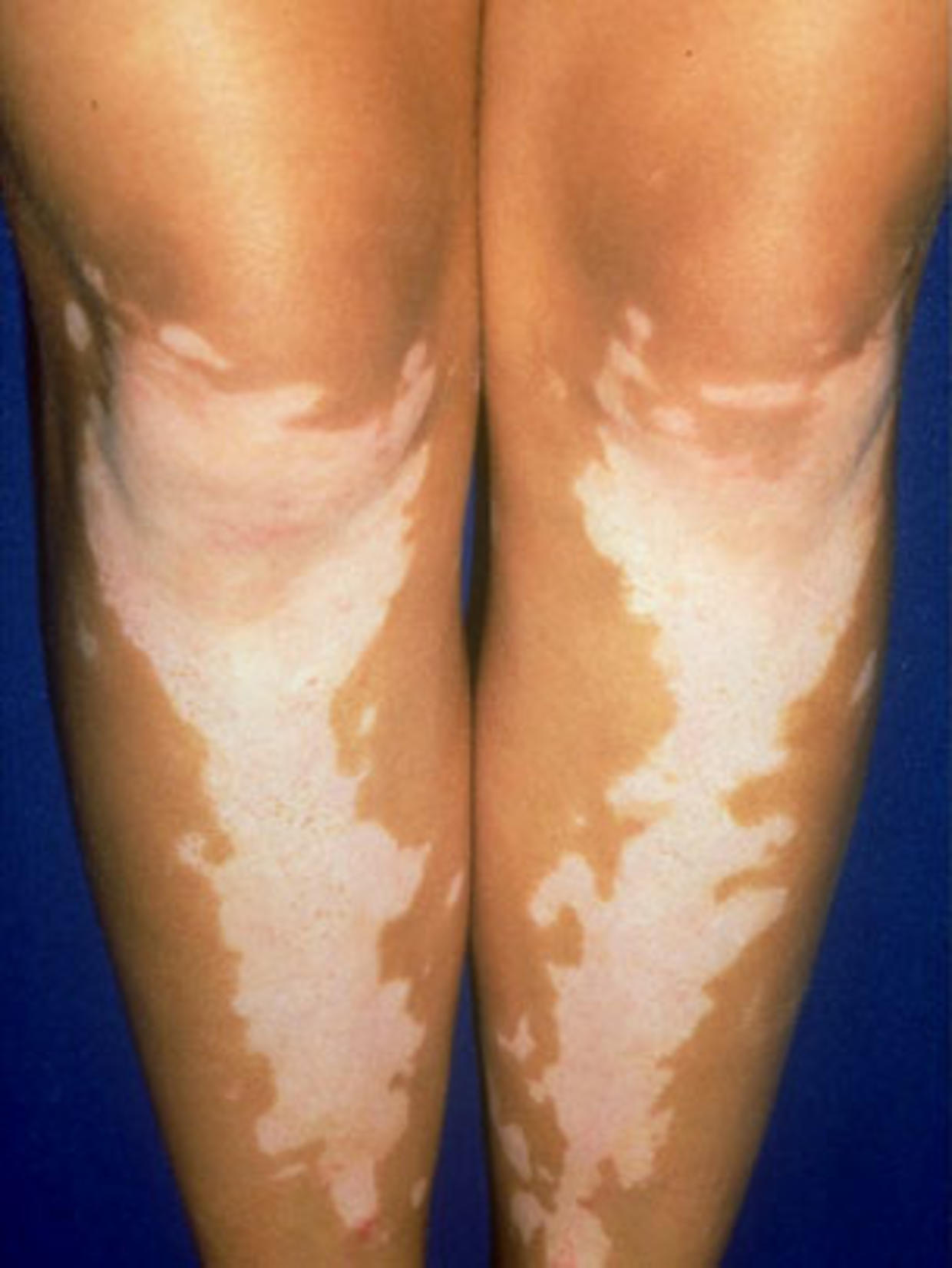
In both trials, subjects with nonsegmental vitiligo were randomized to treatment with Opzelura or placebo cream twice daily for 24 weeks, followed by an additional 28 weeks of treatment with Opzelura for all subjects. Safety and effectiveness of Opzelura were demonstrated in two clinical trials, NCT04052425 and NCT04057573. Many people with vitiligo are also affected by at least one other autoimmune disorder. In people with vitiligo, the immune system appears to attack the pigment cells (melanocytes) in the skin. Vitiligo is considered to be an autoimmune disorder. Depigmentation may occur on the face, neck, and scalp, and around body openings such as the mouth and genitals, as well as areas that tend to experience rubbing or impact, such as the hands and arms. The condition involves loss of pigment (depigmentation) in patches of skin. Nonsegmental vitiligo is the most common form of vitiligo. Satisfactory patient response may require treatment with Opzelura for more than 24 weeks. Opzelura is applied twice a day to affected areas of up to 10% of the body’s surface area. Opzelura is the first FDA-approved pharmacologic treatment to address repigmentation in vitiligo patients. Opzelura is a topical Janus kinase (JAK) inhibitor currently approved for the topical short-term and non-continuous chronic treatment of mild to moderate atopic dermatitis in non-immunocompromised patients 12 years of age and older, whose disease is not adequately controlled with topical prescription therapies or when those therapies are not advisable. 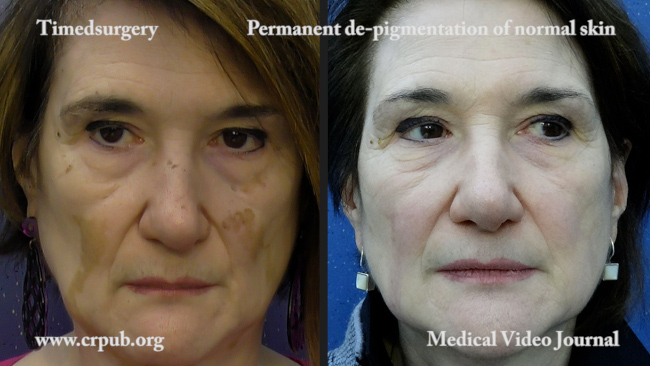
FDA has approved Opzelura (ruxolitinib) cream for the treatment of nonsegmental vitiligo in adult and pediatric patients 12 years of age and older. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
